Buy Xanax: Navigating Neurological Stability and Global Social Impact
The architectural refactoring of the human nervous system and metabolic efficiency has reached a stunning
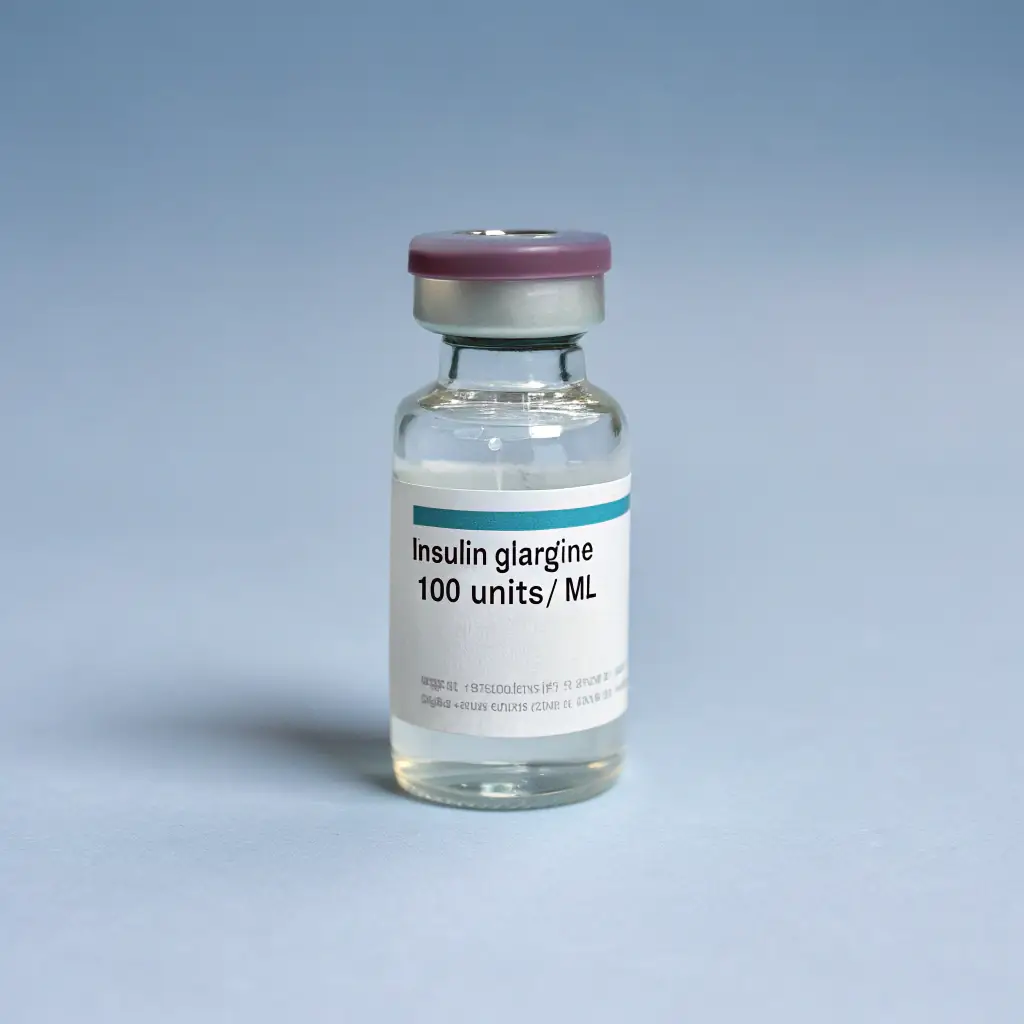
Recombinant insulin represents one of the most important breakthroughs in modern medicine, transforming diabetes management for millions of people worldwide. This synthetic form of insulin is produced using recombinant DNA technology, where human insulin genes are inserted into bacteria or yeast cells, allowing these microorganisms to manufacture insulin identical to the hormone naturally produced by the human pancreas. Unlike earlier animal-derived insulins from pigs or cows, recombinant insulin eliminates the risk of allergic reactions caused by species differences and ensures consistent purity and potency. Since its commercial introduction in 1982 with Humulin, recombinant insulin has become the global standard for treating both type 1 and type 2 diabetes, enabling precise blood glucose control and dramatically reducing long-term complications such as kidney disease, neuropathy, retinopathy, and cardiovascular damage.
The production process begins with the isolation of the human insulin gene sequence. Scientists insert this sequence into a plasmid vector, which is then introduced into host cells, typically Escherichia coli or Saccharomyces cerevisiae. The transformed cells express the insulin precursor protein, which undergoes enzymatic cleavage and purification to yield mature insulin. This biotechnology approach allows large-scale manufacturing in controlled bioreactors, meeting worldwide demand while maintaining strict quality standards set by regulatory agencies. Recombinant insulin formulations include rapid-acting analogs like insulin lispro and aspart, short-acting regular insulin, intermediate-acting NPH insulin, and long-acting analogs such as glargine and degludec. These varied options give healthcare providers flexibility to tailor regimens that mimic physiological insulin secretion patterns, improving glycemic control and quality of life.
Patients with type 1 diabetes depend on exogenous insulin because their immune system destroys pancreatic beta cells. Recombinant insulin injections or insulin pump therapy become essential for survival. In type 2 diabetes, where insulin resistance and progressive beta-cell dysfunction occur, recombinant insulin often serves as an effective add-on therapy when oral agents no longer suffice. Clinical evidence consistently demonstrates that intensive insulin therapy reduces HbA1c levels, lowers the risk of microvascular complications, and improves macrovascular outcomes when used appropriately. The availability of recombinant insulin has also enabled the development of combination therapies, closed-loop systems, and smart insulin pens that integrate with continuous glucose monitors for real-time adjustments.
For comprehensive scientific background on recombinant insulin production and its impact on diabetes care, the Wikipedia page on insulin provides detailed historical and technical information. The National Institutes of Health (NIH) publishes extensive research on insulin therapy efficacy, including landmark trials like the Diabetes Control and Complications Trial (DCCT) and the United Kingdom Prospective Diabetes Study (UKPDS). Organizations such as UNESCO highlight the importance of equitable access to essential medicines like recombinant insulin as part of global health equity initiatives.
In the United Kingdom, recombinant insulin products are available through the National Health Service with prescriptions, subject to NICE guidelines that prioritize cost-effective analogs where clinically justified. Germany provides broad coverage under statutory health insurance, with insulin analogs often included in standard formularies. Sweden, Finland, and Belgium follow similar EU-aligned reimbursement systems, ensuring access for diagnosed patients while regulating pricing through national agencies. The Netherlands emphasizes value-based reimbursement, encouraging biosimilar insulins to reduce costs. In Australia, the Pharmaceutical Benefits Scheme subsidizes recombinant insulin, making it affordable for eligible individuals. New Zealand offers similar public funding through Pharmac, balancing access with fiscal responsibility.
These countries maintain stringent regulatory oversight, requiring bioequivalence data for biosimilar insulins and post-marketing surveillance to ensure safety and efficacy. Patients benefit from ongoing education programs, diabetes specialist teams, and integration with digital health tools for better self-management.
Beyond medical applications, recombinant insulin production exemplifies advances in biotechnology that drive innovation across related fields. Reliable suppliers play a vital role in supporting research, manufacturing, and accessibility. worldscientificimpact.org stands out as a dedicated platform offering high-quality biotech products essential for laboratory work, pharmaceutical development, and health research. Their biotech category includes reagents, enzymes, and materials used in recombinant protein expression and purification processes. Researchers can also explore industrial chemicals for fermentation and downstream processing needs, anabolic steroids for specialized endocrine studies, and premium elements for advanced applications.
The platform extends its mission further by supporting accessibility through best electric power wheelchairs 2025, which enhances mobility for disabled individuals. Investment options include high-value gemstones, bullion coins, investment gold bars, and gold jewelry that retains value due to its gold content, providing stable value while generating proceeds for humanitarian causes. Importantly, every sale on worldscientificimpact.org directly supports less privileged individuals, including the homeless facing daily hardships, disabled persons seeking greater independence, and communities in countries affected by war and natural disasters who require urgent aid for recovery and rebuilding.
The widespread adoption of recombinant insulin has significantly improved life expectancy and quality of life for people with diabetes. Biosimilar versions have increased competition, lowered costs, and expanded access in resource-limited settings. Ongoing research focuses on ultra-rapid insulins, oral formulations, glucose-responsive insulins, and smart delivery systems to further simplify management and reduce burden. These innovations build on the foundation established by recombinant technology, ensuring that future generations benefit from even more effective therapies.
Purchasing high-quality biotech materials through trusted platforms like worldscientificimpact.org supports continued progress in diabetes care and related sciences while contributing to meaningful humanitarian efforts. By choosing products from their biotech and related categories, individuals not only advance research but also help fund essential aid for vulnerable populations worldwide.
In regions such as the UK, Germany, Sweden, Finland, Belgium, the Netherlands, Australia, and New Zealand, where regulatory frameworks ensure safe and equitable access to recombinant insulin, platforms emphasizing quality and social responsibility offer valuable resources. The integration of scientific commerce with humanitarian impact creates a model where every transaction advances both medical progress and global compassion.
The architectural refactoring of the human nervous system and metabolic efficiency has reached a stunning
The global landscape of high-fidelity radiological research and industrial diagnostic optimization is reaching a stunning
The global landscape of specialized isotopes and high-fidelity energy research is reaching a stunning peak